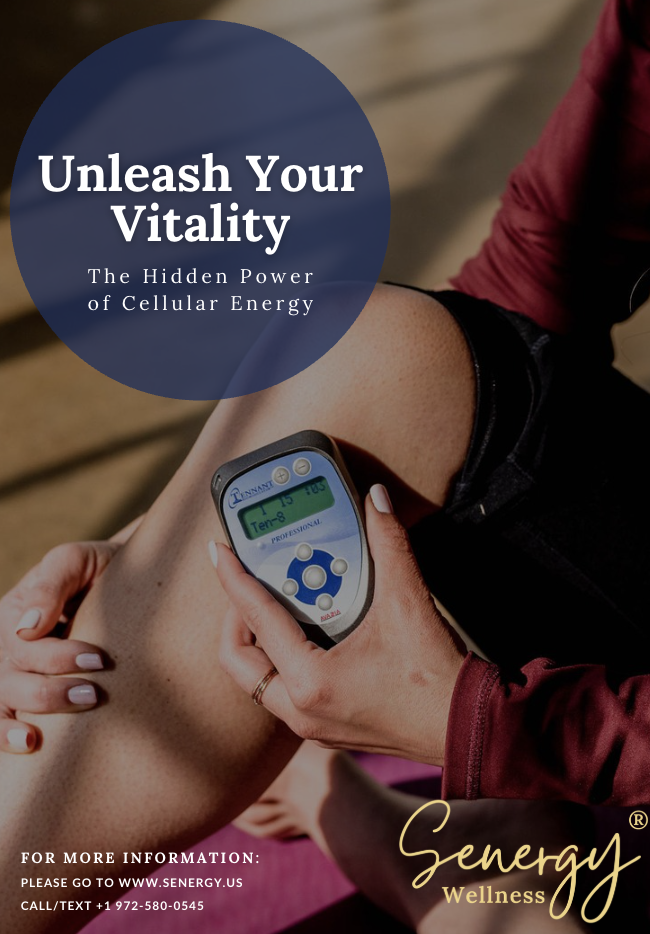
The human body relies on electrical signals to support communication between cells, nerves, and tissues, and those signals play a foundational role in movement, sensation, and internal regulation. Microcurrent technology is designed to interface with these systems using extremely low-level electrical stimulation, which is why many people explore it as a drug-free approach to supporting recovery, circulation, and nervous system balance. The Tennant BioModulator stands out by making this technology practical and accessible in a portable device that can be used at home or in a practitioner setting, making it especially appealing to individuals dealing with chronic discomfort, people seeking alternative wellness strategies, those interested in nervous system regulation or non-pharmaceutical recovery tools, and practitioners looking for a professional microcurrent solution.